Introduction to pH Control in E. coli Fermentation
Table of Contents
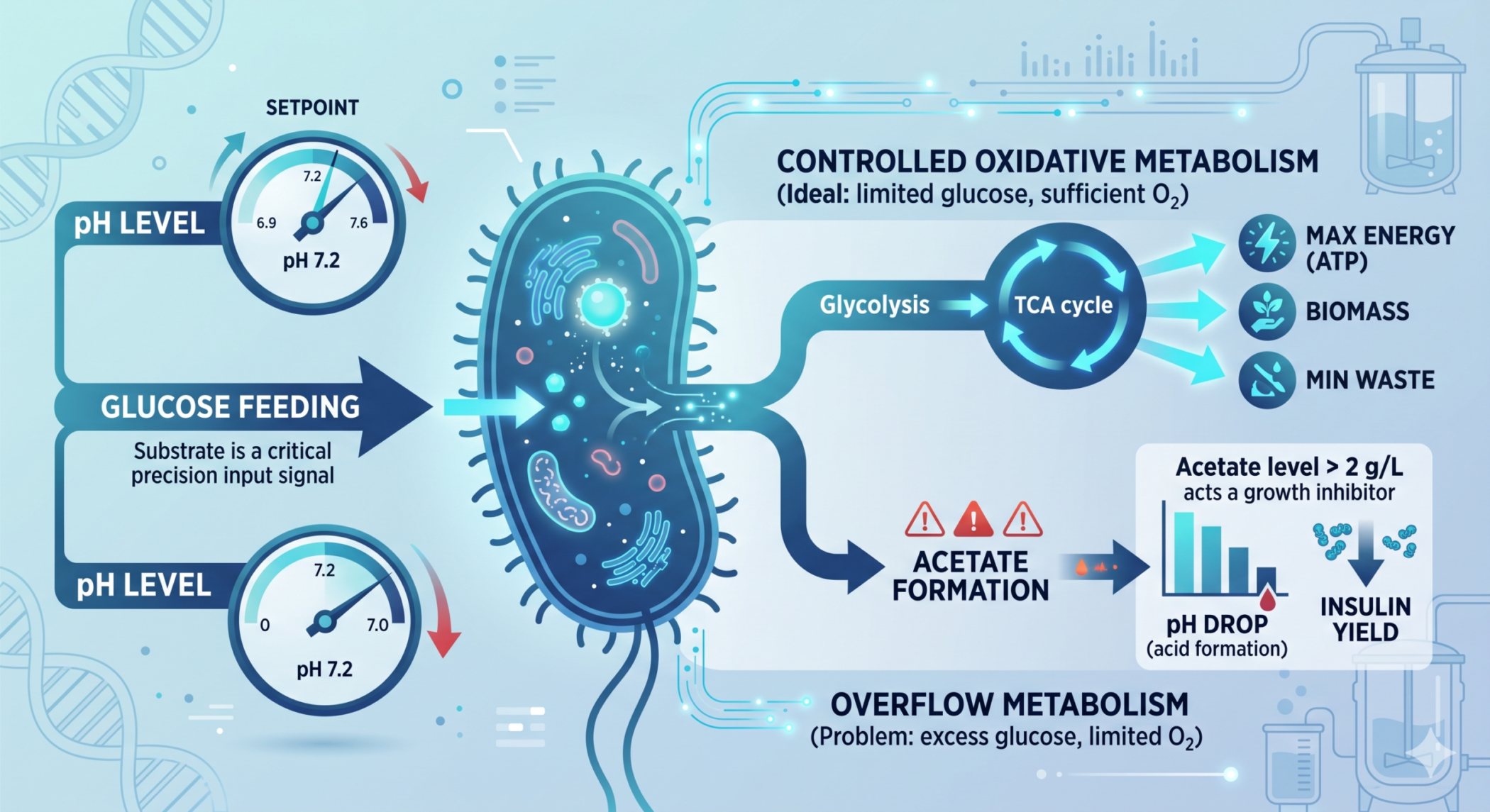
ToggleIn industrial bioprocessing, pH control in E. coli fermentation is one of the most critical factors determining success or failure—especially in recombinant insulin production.
While strain engineering gets attention, real-world performance depends on how well you control the fermentation environment.
Among all process parameters, pH acts as a real-time indicator of cellular metabolism. It reflects:
- Nutrient consumption
- Byproduct formation
- Oxygen availability
- Cellular stress
👉 In simple terms:
pH is the fastest way to understand what your cells are experiencing.
Why pH Control is Critical in Insulin Fermentation
In fed-batch fermentation, even a small deviation in pH can:
- Shift metabolic pathways
- Increase acetate formation
- Reduce protein expression
- Impact downstream processing
📊 Industrial insight:
Acetate levels above 2–5 g/L can significantly reduce insulin yield.
Maintaining pH at 7.2 ± 0.2 ensures:
- Optimal enzyme activity
- Efficient glucose utilization
- Stable protein expression
Understanding pH as a Metabolic Indicator
pH Reflects Cellular Metabolism
During fermentation:
- Rapid glucose metabolism → acid formation → pH decreases
- Ammonia addition (NH₄OH) → pH increases
👉 Interpretation:
- pH ↓ = Acid formation (overfeeding or oxygen limitation)
- pH ↑ = Carbon limitation or reduced metabolism
Link Between pH, Carbon, and Nitrogen
pH is directly linked to:
- Glucose (carbon source)
- Ammonia (nitrogen source)
Balanced system:
- Controlled glucose → stable pH
- Efficient metabolism → low acetate
Imbalanced system:
- Excess glucose → acetate → pH drop
- Low glucose → ammonia dominance → pH rise
Standard Process Conditions
Typical industrial setup:
- Organism: Recombinant E. coli
- Mode: Fed-batch fermentation
- pH: 7.2 ± 0.2
- Temperature: 30–37°C
- Induction: OD > 45
- Carbon source: Glucose
High cell density:
- OD600: 80–100+
Phase-Wise pH Behavior in Fed-Batch Fermentation
1. Batch Phase
- Excess glucose
- Acid formation
- pH decreases
👉 Watch: Rate of pH drop
2. Transition Phase
- Feeding starts
- pH stabilizes
👉 Deviations:
- pH ↓ → Overfeeding
- pH ↑ → Underfeeding
3. Mid Fed-Batch Phase
- Efficient metabolism
- Low acetate
- Slight pH rise
4. Pre-Induction Phase
- Highly sensitive
- Oxygen demand peaks
👉 Critical signals:
- pH drop → Oxygen limitation
- pH rise → Carbon limitation
5. Induction Phase
- Growth slows
- pH rises slightly
👉 Key action:
- Reduce feed by 30–50%
6. Late Phase
- Metabolism slows
- pH drifts upward
Industrial pH Control Strategy
Base and Acid Selection
- NH₄OH (Base):
- Controls pH
- Provides nitrogen
- H₃PO₄ (Acid):
- Fine pH correction
- Provides phosphate
👉 Important:
You are feeding + controlling at the same time.
PID and Cascade Control
Advanced systems use:
- PID loops
- Cascade control (pH + DO + feed)
Poor tuning leads to:
- Oscillations
- Overcorrection
- Process instability
Advanced Interpretation Using pH
Base Consumption Rate (BCR)
BCR is a powerful soft sensor:
- High BCR → Active metabolism
- Stable BCR → Balanced system
- Low BCR → Stress or limitation
Detecting Acetate Formation Early
Early indicators:
- Rapid pH drop
- Increased BCR
- Stable DO
👉 Action:
- Reduce feed
- Increase oxygen transfer
Integrated Control: pH vs DO vs Feed
Decision Logic Used in Industry
| Observation | Interpretation | Action |
|---|---|---|
| pH ↓ + DO ↓ | Oxygen limitation | Increase aeration |
| pH ↓ + DO ↑ | Overfeeding | Reduce feed |
| pH ↑ + Low BCR | Underfeeding | Increase feed |
| pH ↑ + High BCR | Control issue | Tune PID |
👉 This is the core of industrial fermentation control.


Real Industrial Example (5 KL Fermenter)
Inputs:
- Volume: 5000 L
- Biomass: 20 g/L
- µ: 0.15 hr⁻¹
- Yield: 0.45
- Maintenance: 0.03

≈ 72–73 L/hr (pre-induction)
After induction:
≈ 28 L/hr
Common Mistakes in pH Control
- Overfeeding glucose
- Ignoring oxygen limitation
- Poor PID tuning
- Relying only on pH (no correlation)
Expert Tips for High Cell Density Fermentation
- Always analyze trends, not snapshots
- Avoid aggressive corrections
- Link feed with metabolism
- Use pH as a predictive tool
Golden Rule:
Fix metabolism → pH stabilizes automatically
Conclusion
Mastering pH control in E. coli fermentation is not about maintaining a number—it’s about understanding the biology behind it.
By integrating:
- pH
- DO
- Feed rate
- Metabolic signals
You can shift from:
👉 Reactive control → Predictive optimization
This is what defines world-class insulin fermentation processes.
FAQs
1. Why is pH maintained at 7.2 in E. coli fermentation?
It provides optimal conditions for growth, enzyme activity, and minimizes acetate formation.
2. What causes pH drop during fermentation?
Acid formation due to overfeeding or oxygen limitation.
3. Why is NH₄OH used for pH control?
It acts as both a base and a nitrogen source.
4. How is glucose linked to pH?
Glucose metabolism produces acids, directly affecting pH.
5. Why reduce feed after induction?
To reduce metabolic burden and improve protein expression.

 Golden Rule:
Golden Rule: