In biopharmaceutical manufacturing, Steam-in-Place (SIP) in Bioprocessing is not just an objective—it is a strict requirement. Whether operating upstream fermentation systems or downstream purification equipment, maintaining a contamination-free environment is critical.
Steam-in-Place (SIP) is one of the most important sterilization techniques used to ensure equipment sterility without dismantling process systems. When properly designed and executed, SIP safeguards product quality, ensures regulatory compliance, and prevents costly batch failures.
This guide explains SIP from a practical, real-world engineering perspective—clear, structured, and directly applicable to shop-floor operations.
Understanding the Role of SIP
Table of Contents
ToggleSteam-in-Place (SIP) is a sterilization method that uses saturated clean steam to eliminate microorganisms from process equipment such as:
- Bioreactors and fermenters
- Process piping
- Storage tanks
- Filtration systems
Unlike manual sterilization, SIP is integrated into the system design, allowing sterilization without disassembly. This reduces downtime, minimizes contamination risks, and improves operational efficiency.
In regulated environments aligned with FDA and EMA expectations, SIP helps achieve a Sterility Assurance Level (SAL) of 10⁻⁶, meaning the probability of a surviving microorganism is extremely low (less than one in a million).
Important principle: SIP must always follow Clean-in-Place (CIP).
Residual dirt, protein, or biofilm can protect microorganisms and reduce sterilization effectiveness.
Why Steam is Highly Effective
The effectiveness of SIP comes from the latent heat of vaporization of steam.
When steam contacts a cooler surface, it condenses into water and releases a large amount of heat energy instantly. This rapid heat transfer destroys microorganisms, including heat-resistant spores.
Key Requirements for Effective SIP:
- Use saturated steam (not superheated steam)
- Ensure steam is free from non-condensable gases (NCGs) such as air
Even small amounts of trapped air can act as insulation, reducing heat transfer and creating cold spots, which compromise sterilization.
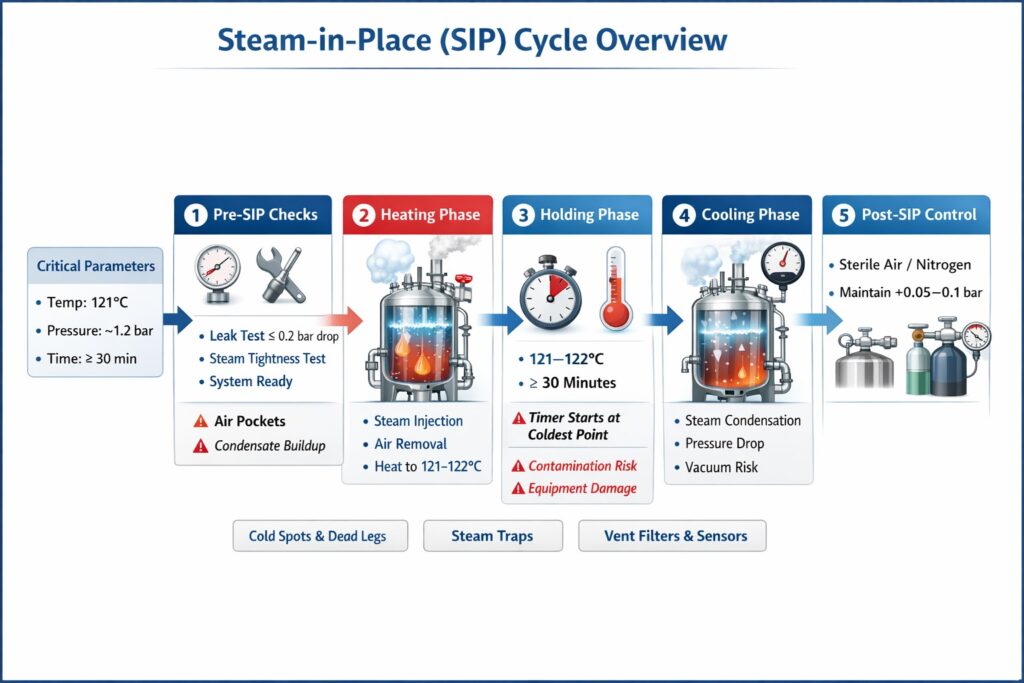
Critical SIP Parameters (The Core Three)
Every SIP cycle is controlled by three essential parameters:
Parameter | Typical Range | Purpose |
Temperature | 121–122°C | Microbial and spore destruction |
Pressure | ~1.0–1.2 bar(g) | Maintains steam conditions |
Time | ≥ 30 minutes | Ensures complete sterilization |
These values are established through thermal validation and biological indicators.
Pre-SIP: System Integrity Verification
Before introducing steam, the system must be completely leak-tight.
Pneumatic Pressure Test
- Pressurize system to ~1.5 bar
- Hold for 10 minutes
- Acceptance: Pressure drop ≤ 0.2 bar
Failure indicates:
- Leaking gaskets
- Improper valve sealing
- Loose fittings
Even minor leakage can lead to contamination during cooling.
Steam Tightness Test
- Conducted at ~2 bar using steam
- Duration: 5–10 minutes This ensures the system can withstand thermal expansion and real steam conditions.
Vent Filters: Critical Sterility Barrier
Vent filters (commonly hydrophobic PTFE) prevent contamination from entering the system during pressure changes.
Key Control Parameter:
- Differential pressure ≤ 70 mbar
If exceeded:
- Filter membrane may be damaged
- Pore blockage (blinding) may occur
Routine Integrity Tests:
- Bubble point test
- Diffusion test
A failed vent filter directly compromises sterility.
SIP Cycle Phases
A) Heating Phase
Objective: Raise the entire system to sterilization temperature.
Challenges:
- Air entrapment
- Uneven steam distribution
- Condensate accumulation
Best Practices:
- Open vents to remove air
- Ensure proper drainage
- Maintain sloped piping
B) Holding Phase
This is the actual sterilization step.
- Temperature: 121–122°C
- Pressure: ~1.2 bar
- Time: Minimum 30 minutes
Critical Rule:
Holding time begins only when the coldest point reaches target temperature.
This is why temperature mapping is essential during validation.
C) Cooling Phase
This phase is often underestimated but highly critical.
When steam condenses:
- Large volume reduction occurs
- Internal pressure drops rapidly
- Vacuum is created inside the system
Risks:
- Equipment deformation
- Air ingress (contamination)
Cold Spots: A Major Sterility Risk
Cold spots are areas that fail to reach sterilization temperature.
Common Locations:
- Dead legs
- Long pipe branches
- Valve bodies
- Filter housings
- Low-point drains
Temperature Sensor Placement:
Install sensors at:
- Lowest points (drain locations)
- Farthest points from steam entry
If these locations meet sterilization criteria, the system is considered compliant.
Condensate Management
Steam sterilization depends on condensation—but excess condensate creates problems.
Issues Caused by Condensate:
- Blocks steam flow
- Reduces heat transfer efficiency
- Creates cold spots
- Damages filters
Solution: Steam Traps
Steam traps remove condensate while retaining steam.
Common Types:
Thermodynamic Trap
- Works on velocity and pressure difference
- Compact and robust
Best for:
- High-pressure systems
- Main steam lines
Mechanical Trap
- Uses float or bucket mechanism
Example:
- Float rises with condensate → opens valve
Best for:
- Continuous condensate removal
- Stable systems
Thermostatic Trap
- Works on temperature difference
Opens when temperature is low (condensate present)
Closes when steam temperature is reached
Best for:
- Air venting
- Startup conditions
Good Design Practices:
- Proper piping slope
- No stagnant zones
- Adequate drainage points
Post-SIP: Managing Vacuum Formation
During cooling:
- Steam condenses rapidly
- Internal pressure decreases
- Vacuum forms inside equipment
Why This is Dangerous:
- Can collapse vessels
- Can pull contaminated air inside
- Can damage seals and gaskets
Solution: Maintain Positive Pressure
Introduce sterile gas such as:
- Sterile air
- Nitrogen (N₂)
Recommended Practice:
- Maintain slight positive pressure (0.05–0.1 bar)
Continuously monitor via control system
Automation and Control Systems
Modern SIP systems are operated through Distributed Control Systems (DCS).
Key Functions:
- Recipe-based operation
- Temperature and pressure control
- Alarm handling
- Data recording for compliance
Benefits:
- High repeatability
- Reduced operator dependency
- Full traceability for audits
Final Thoughts
Steam-in-Place is more than just a sterilization step—it is a critical control strategy that directly impacts product safety and batch success.
A robust SIP system depends on:
- Proper system design
- Understanding of thermodynamics
- Effective monitoring and validation
When implemented correctly, SIP becomes a reliable and invisible process—ensuring sterility without interruption.
Best Practices for Reliable SIP:
Based on real plant experience:
- Validate cold spots, not average temperature
- Ensure complete air removal before heating
- Maintain high-quality saturated steam
- Regularly test filters and steam traps
- Monitor pressure during cooling
- Avoid dead legs beyond 1.5D rule
Example: If your main pipe diameter (D) = 100 mm
Therefore, Maximum dead leg length (L) allowed:
L≤1.5×100=150 mm
FAQs
- What is the standard SIP condition?
Typically 121–122°C for at least 30 minutes, depending on system validation.
- Why is saturated steam required?
Because it provides efficient heat transfer through condensation, ensuring effective sterilization.
- What causes cold spots?
Air pockets, poor drainage, dead legs, and improper steam distribution.
- How is vacuum prevented after SIP?
By introducing sterile air or nitrogen to maintain positive pressure.
- What is the acceptable leak test limit?
Pressure drop should not exceed 0.2 bar in 10 minutes at 1.5 bar.